Claritas NucMed Technologies Ltd has received approval from the Australian Therapeutic Goods Administration (TGA) for its innovative medical software device, Claritas iPETTM. This significant clearance enables the supply of the software in Australia, marking an important milestone for the healthcare technology company that specializes in advanced imaging solutions.
With this approval, Claritas iPETTM expands its reach, already having been cleared for use in several other jurisdictions. The software is designed to enhance the quality of Positron Emission Tomography (PET) scans, facilitating clearer and sharper images. It integrates seamlessly into existing hospital systems, regardless of the equipment type or manufacturer, making it a versatile tool for healthcare providers.
Enhancing Diagnostic Imaging
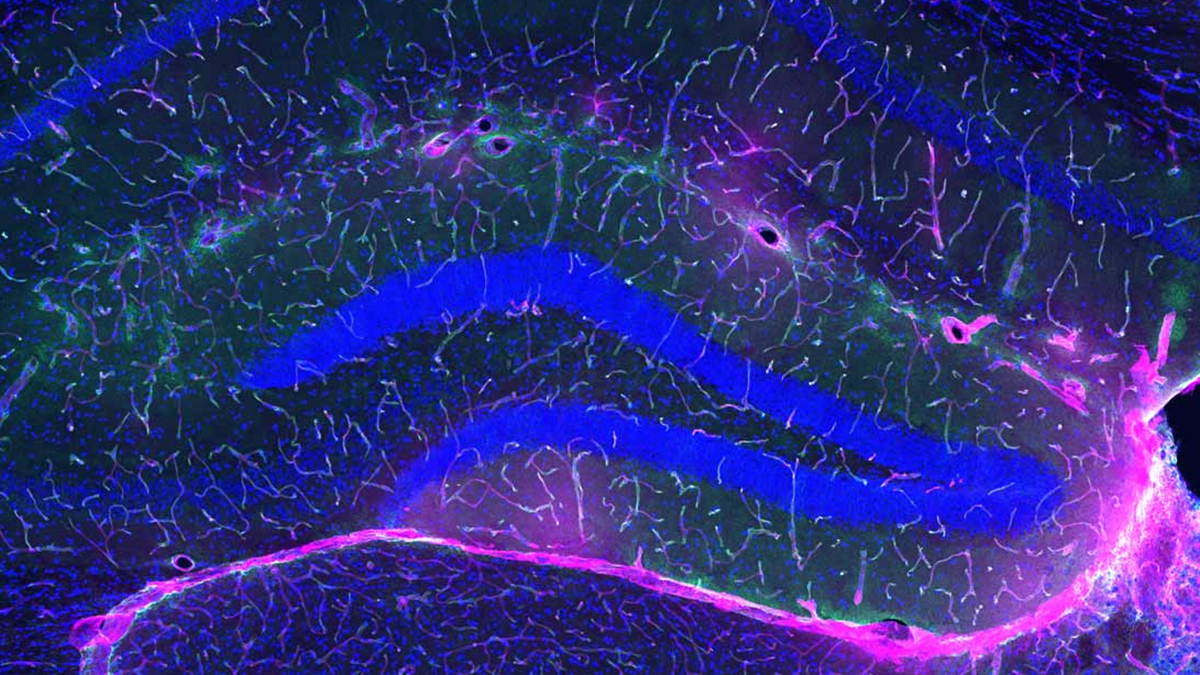
The primary function of Claritas iPETTM is to improve the visibility of PET scans by reducing noise and enhancing image clarity. It ensures that fine lesions are clearly visible, maintains standardized uptake values (SUV), and does not introduce artifacts during processing. This capability is particularly beneficial for hospitals and clinics aiming to optimize scan throughput and enhance patient experience.
By allowing for reduced scan times and lower isotope dosages, the software contributes to a significant decrease in radiation exposure for patients. Moreover, the integration of computed tomography (CT) or magnetic resonance imaging (MRI) data as overlays on PET scans provides additional clarity and information, enhancing diagnostic accuracy.
Dr. Fernando Salis, a nuclear medicine physician and director of Claritas NucMed Brazil, expressed satisfaction with the software’s implementation in Brazil, noting its impact on improving the quality and efficiency of PET scans. He highlighted that hospitals utilizing Claritas iPETTM reported an increase in the number of patients scanned daily, which not only improves access but also reduces costs.
Commitment to Innovation and Compliance
Claritas NucMed Technologies Ltd, a subsidiary of Claritas HealthTech Ltd, is headquartered in London, United Kingdom. The company is the authorized distributor of Claritas iPETTM alongside other nuclear medicine devices, including iPETcertumTM and iSPECTcardiacTM. Their research and development are focused on image enhancement and artificial intelligence technologies aimed at transforming the diagnostics landscape.
All products from Claritas are designed to comply with international regulations, including HIPAA, DPDPA, and GDPR standards, ensuring that they meet the highest standards of data protection and patient privacy. For further details, potential users and partners can visit www.claritashealthtech.com.
This TGA approval underscores Claritas‘ commitment to enhancing medical imaging capabilities and improving patient outcomes across the globe.